Proteins and Cancer
In every cell in the human body, there are countless tiny molecules, called proteins, that work together to allow the cell to harvest energy, move, reproduce, and interact. In cancer cells, some proteins can malfunction or be functioning out of control, causing the cells to grow abnormally and threaten normal cells.
Targeting p97
P97 is one such protein that gives cells in some cancer types a growth advantage. Researchers have been developing compounds to regulate p97, but it was not clear how they worked. In a paper published in Science (1), researchers from Sriram Subramaniam’s lab at the National Cancer Institute were able to visualize the binding of a drug inhibitor to p97 by using high-resolution single particle cryo-electron microscopy (cryo-EM), an advanced imaging technique.
A Wrench in the Works
Normally, when p97 is activated, it moves in two steps (also visualized by cryo-EM for the first time in this paper!). The bottom, D2 domain (purple in the above image) moves, then the top two domains, the D1 and N (blue and green), move. However, when the inhibitor was added, it stuck to p97 between the D2 and D1 domains, and got in the way of the first step. Like a wrench jammed into gears, the drug keeps p97 from working.
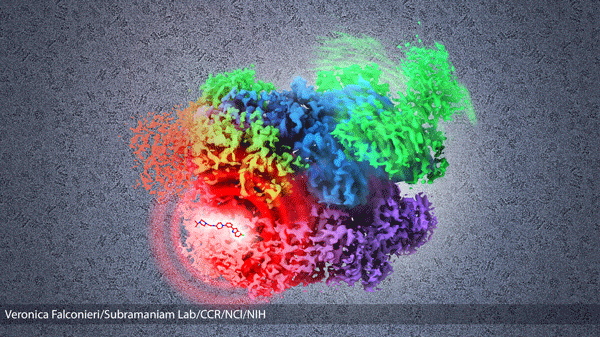
Inhibition of cancer protein p97: This illustration shows a composite structure derived from 5 cryo-EM structures of cancer protein p97 – no activators bound, activators (ATP molecules) bound (3 structures), and inhibitor bound structures. The inhibitor bound structure is locked in the inactive shape (left), and prevented from progressing through its usual dynamic conformation change when exposed to activators (right). Protein images taken through the cryo-electron microscope fill the background. (Veronica Falconieri/Subramaniam Lab/CCR/NCI/NIH)
Smarter Design
Now that scientists know how the inhibitor works, they can design inhibitor variations that may be better for pharmaceutical applications. The end result of this research will hopefully be a more effective drug to fight against cancer types that use p97 abnormally to grow.
Links
References
- Banerjee, A. Bartesaghi, A. Merk, P. Rao, S. L. Bulfer, Y. Yan, N. Green, B. Mroczkowski, R. J. Neitz, P. Wipf, V. Falconieri, R. J. Deshaies, J. L. S. Milne, D. Huryn, M. Arkin, and S. Subramaniam, “2.3 Å resolution cryo-EM structure of human p97 and mechanism of allosteric inhibition,” Science, Jan. 2016.